
Alma Diagnostic Services
Alma pharmaceutical Diagnostic Services offer clients a range of Genomic Services from multiple platforms, generating high quality data from DNA and RNA.
As part of our Genomic Service offering Alma pharmaceutical can also provide a premium ‘white glove’ Specialty Central Lab service on a ‘Project by Project’ basis. We can take care of your end-to end Sample Management needs acting as your central lab vendor who can also seamlessly integrate with other labs.
We can conduct your specimen lab management & processing, storage, data needs and downstream analysis either in-house or work with other vendors to ship samples to them for analysis.
Our specialty lab solutions include a range of qPCR and NGS platforms with a range of proteomic and single cell options to aid with a ‘multi-omics’ approach.
Our dedicated Data Sciences team has developed proprietary, innovative, bioinformatics solutions. Helping clients generate better insights from genomic data to uncover novel research findings that aid advancements in drug development.

Pharmaceutical Drug Product Development
Addressing the increasing pressure on pharmaceutical companies to bring clinical candidates through their pipeline faster and with greater efficiency, we provide expert pharmaceutical drug product development and manufacturing solutions. Working in true partnership we will support your solid oral dose product development programme through experience, expertise, and efficiency
Operating from state-of-the-art, custom designed facilities, our scientists can develop your clinical candidates into optimum formulations and manufacture solid oral dose products for all stages of clinical trials.
From developing a fit-for-purpose formulation for First-in-Human trials, to scaling up for late phase trials, registration and ultimately scaling up for ongoing commercial supply, our pharmaceutical drug product development solutions are tailored to meet your needs.
Our purpose-built facilities in Craigavon and at the Charnwood Campus, UK provide solid oral dose drug product development, manufacturing and analytical testing capacity, ensuring we can meet your future pharmaceutical development and manufacturing needs..

API Services & Chemical Development
Developing a small molecule API (active pharmaceutical ingredient) involves undertaking various chemical processes to ensure that the finished product meets the stringent standards for identity, quality, purity and strength required by regulatory authorities. Small molecule API development and manufacturing, therefore, can be a complex process, particularly when considering scale-up.
Alma’s strength in drug substance API development and manufacture is proven by being the pharma CDMO partner of choice for many pharma and biotech companies seeking integrated drug development solutions from molecule to market. Our technical expertise in complex API development, scale-up and manufacture, coupled with our extensive facilities enable us to offer integrated API contract manufacturing solutions from early phase clinical development to commercial API manufacture, which are fully supported by dedicated analytical and regulatory teams.

Early Phase Pharmaceutical Drug Product Development
Alma’s experienced formulation development scientists can develop a range of oral dose formulations to support your early phase clinical trials.
With both non-GMP and GMP facilities, we offer flexible and efficient solutions to help you develop a fit-for-purpose formulation and manufacture early phase clinical materials.
Preclinical or Phase 1 First-in-Human solutions
With a proven capability of handling potent APIs and peptide pharmaceutical drug programmes, our full suite of Xcelodose, All-Fill and specialised technologies help accelerate timelines whilst maintaining high quality.

Specialised Support Service
Spectroscopy is critical within analytical drug development and is used to identify compounds and verify purities / impurities.
With a wide range of sophisticated MS and NMR instruments, Almac provides method development and validation of methods for identification of unknowns, genotoxic impurities, elemental impurities and other requirements. Almac’s specialist team has over 15 years’ experience in solving clients’ problems.
Our scientists are highly experienced in the development of high quality assays using state-of-the-art LC-MS/MS technology – Applied Biosystems API 3000/4000 instrumentation which incorporates the automated reporting of data. This specialist analyst software is compliant with FDA 21 CFR Part 11.
We excel in the development and validation of routine and non-routine methods in a very wide range of matrices ( i.e. muscle, kidney, liver, fat, bone, bone marrow, synovial fluid, cardiac tissue, plasma, serum, whole blood, urine and faeces) and apply these methods in a GLP and GCP compliant environment.
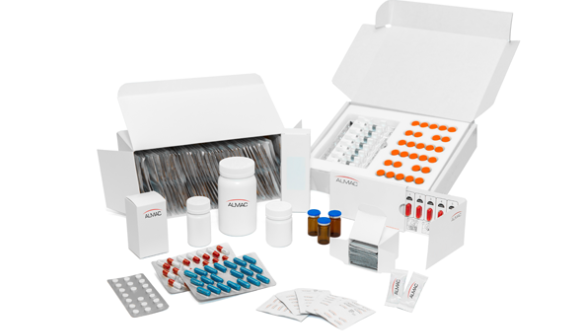
Commercial Packaging
With commercial facilities in the UK, EU & US, our global presence and local expertise provides flexible, quality led commercial pharmaceutical packaging solutions tailored to meet your specific needs, from initial artwork and pack design to commercial drug product packaging, serialisation and distribution.
We are MHRA, HPRA and FDA licensed and have been successfully inspected by global regulatory authorities including Pmda, ANVISA, Turkish Ministry of Health and South Korea FDA. With DEA and UK Home Office approval we are also licensed for the storage and processing of controlled drugs (schedules II-V). In line with regulatory compliance, our facilities regularly undergo GMP inspections by MHRA, FDA, and HPRA. We hold GMP certificates enabling GMP formulation development, manufacture, and packaging of products, as well as flexible GMP specific floorspace. Pre-qualified amenities within our floorspace include, power, compressed air, purified water, and RH control. Through comprehensive standard operating procedures, rigorous training, and a range of process controls, we can ensure that your products meet the required specifications.
Our high and low throughput commercial pharmaceutical packaging operations provide flexibility and efficiency in the processing of solid oral dose and sterile presentations. We’re able to support both high volume marketed products and niche/orphan drug launches.
We are pleased to offer you the chance to have the healthy
Meet Our Specialist
This strategy is set, driven and overseen by our Board of Directors and Senior Executive team to ensure it plays an integral part in all company operations. Each day, the tactical aspects of this strategy are demonstrated, upheld and nurtured by our diligent management and operational teams.

With access to
24 Hour
Emergency
Assistance
Addressing increased client demand for developing and bringing more advanced medicines and niche orphan drugs to market, we enhanced our commercial pharmaceutical packaging offering by adding bespoke technology for the specialised packaging of complex kit assembly for medical devices, combination products and biologic packs.







